Overview & Epidemiology
Lumbar disc herniation (LDH) is one of the most common and clinically significant conditions in orthopaedic and spinal surgery, affecting approximately 1–3% of the population at any given time and accounting for the majority of surgically treated spinal conditions worldwide. It occurs when nuclear material from the intervertebral disc — the nucleus pulposus — migrates through a defect in the annulus fibrosus to compress adjacent neural structures (nerve roots or, in severe cases, the cauda equina). The clinical syndrome that results — radiculopathy — is characterised by pain, paraesthesia, and neurological deficit in the dermatomal and myotomal distribution of the compressed nerve root, and must be distinguished from referred (non-dermatomal) back pain. The vast majority of lumbar disc herniations (approximately 90%) resolve with conservative management within 6–12 weeks; surgery is indicated for a minority of patients who fail conservative management or who present with progressive neurological deficit or cauda equina syndrome.
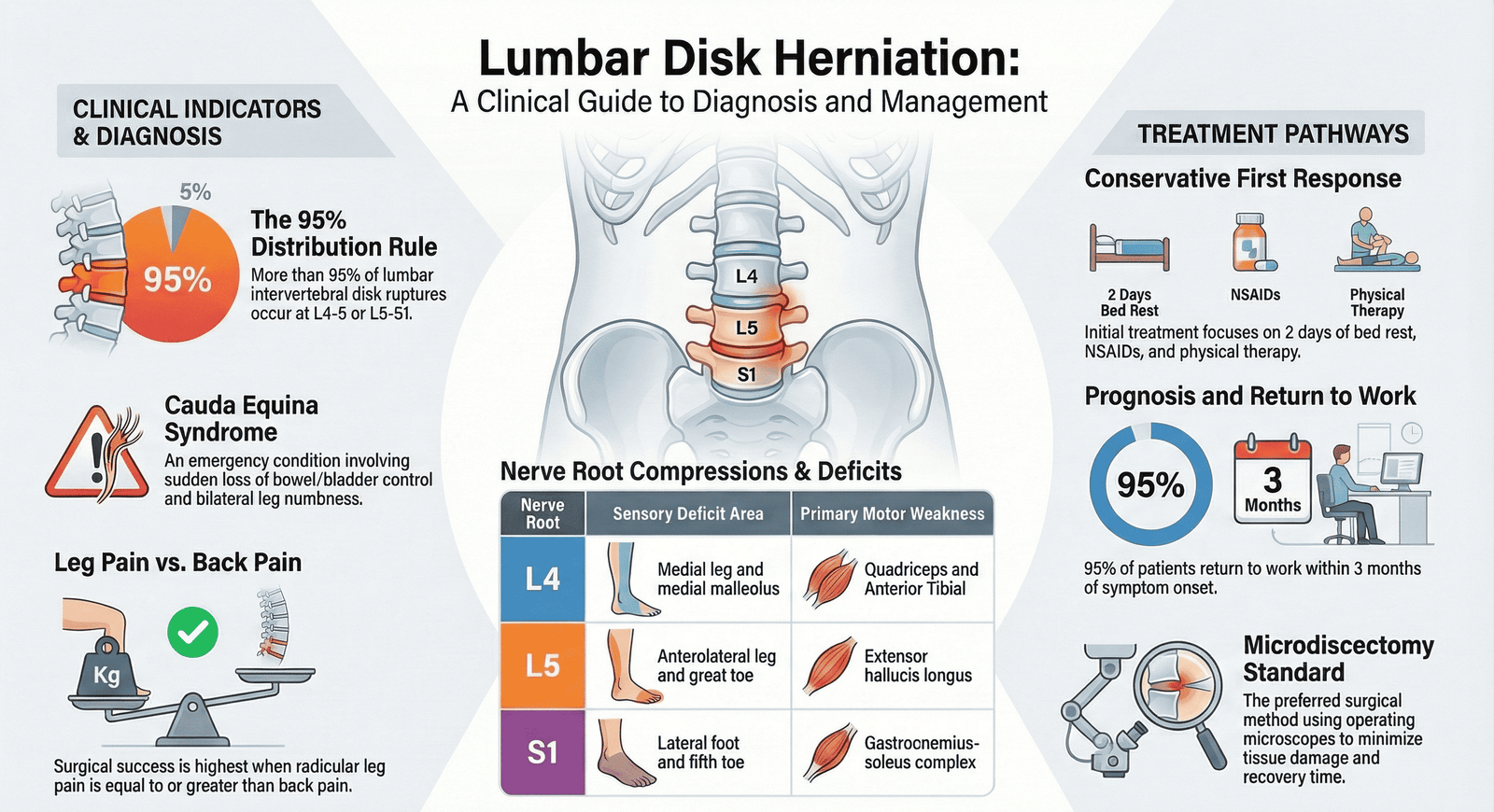
- Epidemiology: peak incidence at 30–50 years; male predominance (male:female ratio approximately 2:1); the L4/5 and L5/S1 levels account for approximately 90–95% of all symptomatic lumbar disc herniations; L3/4 accounts for approximately 5%; higher lumbar levels (L1/2, L2/3) are uncommon; the most common root compressed at L4/5 is L5; at L5/S1 is S1; at L3/4 is L4
- Risk factors: heavy physical labour and repetitive bending/twisting; prolonged sitting (truck drivers, office workers); smoking (impairs disc nutrition); obesity; previous disc herniation; genetic predisposition (familial aggregation of disc degeneration has been demonstrated); age (disc water content decreases progressively after the third decade, reducing the nucleus`s ability to absorb and distribute compressive loads); sudden heavy lifting or Valsalva manoeuvre precipitates acute herniation in a predisposed disc
Anatomy — The Intervertebral Disc
- Structure of the intervertebral disc: (1) Nucleus pulposus — the central gelatinous core; composed primarily of type II collagen, aggrecan (a proteoglycan that binds water), and water (approximately 80% in a young adult, falling to 70% with age); the nucleus acts as a hydrostatic pressure vessel that distributes compressive load uniformly across the annulus; the notochordal cells that originally form the nucleus are replaced by fibrochondrocytes with ageing; (2) Annulus fibrosus — the outer fibrous ring; composed of concentric lamellae of type I collagen fibres arranged in alternating oblique directions (approximately ±30° to the disc plane); the outer annulus is innervated and vascularised; the inner annulus is avascular and aneural; the posterior annulus is thinner and weaker than the anterior annulus, explaining the predominance of posterior and posterolateral herniations; (3) Cartilaginous end plates — hyaline cartilage interfaces between the disc and the vertebral body; the primary route of nutrient delivery to the avascular disc by diffusion
- Disc nutrition and degeneration: the disc is the largest avascular structure in the body; nutrition (glucose, oxygen) diffuses from the vertebral end plate capillaries through the end plate cartilage and into the disc; waste products (lactate, CO₂) diffuse in the opposite direction; this diffusion gradient is dependent on end plate integrity and disc hydration; smoking, diabetes, and end plate calcification impair diffusion and accelerate disc degeneration; the `cascade` of disc degeneration: loss of proteoglycan → loss of water content → loss of disc height → increased annular stress → annular tears → herniation or disc collapse
- The posterior longitudinal ligament (PLL): runs along the posterior aspect of the vertebral bodies and intervertebral discs within the spinal canal; the PLL is widest and strongest centrally, tapering laterally; this anatomy explains why most disc herniations occur POSTEROLATERALLY (at the lateral edge of the PLL where it is thinnest and weakest) rather than centrally; a central herniation must rupture through the thicker central PLL and tends to produce central spinal canal compromise (bilateral or cauda equina symptoms) rather than unilateral root compression
Morphological Classification of Disc Herniation
The North American Spine Society (NASS), American Society of Spine Radiology (ASSR), and American Society of Neuroradiology (ASNR) published a consensus classification of disc pathology (2001, revised 2014) that provides standardised terminology for describing lumbar disc herniations on imaging.
| Term | Definition | Morphology on MRI | Clinical Significance |
|---|---|---|---|
| Normal disc | No extension of disc material beyond the interspace; disc material contained entirely within the disc space bounded by the vertebral end plates and the outer annulus | T2-weighted MRI: high signal nucleus (water content); uniform disc height; intact annular margins; no extension beyond vertebral body margins | No neural compression; no symptoms attributable to disc pathology |
| Disc bulge | Generalised or symmetric extension of disc material BEYOND the disc space margin by more than 25% of the disc circumference; the disc extends symmetrically in all directions (like a tyre that is losing air pressure); NOT a herniation; due to laxity of the annular fibres rather than a focal defect; a normal variant in older patients | Smooth, symmetric, circumferential extension of disc margin beyond the end plates; no focal component; the disc appears broad-based and flat | A disc bulge ALONE is a common incidental finding and is NOT the cause of radiculopathy; may contribute to central canal stenosis in combination with other degenerative changes (facet hypertrophy, ligamentum flavum hypertrophy); must be distinguished from focal protrusion |
| Disc protrusion | A FOCAL extension of disc material beyond the disc space involving LESS THAN 25% of the disc circumference; the base of the herniation (at the disc space) is WIDER THAN THE HEIGHT or WIDTH of the herniated material; the annular fibres are disrupted but the outermost annular fibres remain intact (the herniation has NOT completely ruptured through the annulus) | Focal, asymmetric protrusion of disc material; the base is wider than the apex on axial MRI; continuous signal with the parent disc; typically posterolateral in location | The most common type of disc herniation; produces focal nerve root compression; accounts for the majority of radiculopathy presentations; responds well to conservative management; the nucleus remains contained within the outer annulus |
| Disc extrusion | A focal extension of disc material where the HEIGHT or WIDTH of the herniated material is GREATER THAN THE BASE at the disc space; the nucleus has completely ruptured through the full thickness of the annulus but remains CONNECTED to the parent disc by a `neck`; the herniated material may migrate cranially or caudally within the epidural space; the posterior longitudinal ligament may still contain the herniation | The herniated fragment is larger at its apex than at its base on axial MRI; a `mushroom` or `teardrop` shape; may be seen to migrate cranially or caudally on sagittal sequences; remains in continuity with the parent disc | More severe nerve root compression than protrusion; less likely to resolve spontaneously than protrusion (the extruded fragment is larger and causes more displacement of the nerve root); may produce more severe neurological deficit; paradoxically, extrusions may undergo faster spontaneous resorption than protrusions because the exposed nuclear material triggers a greater inflammatory and immunological response (phagocytosis by macrophages) |
| Disc sequestration (free fragment) | The herniated disc material has LOST ALL CONTINUITY with the parent disc — a free fragment of nuclear material lies within the epidural space; the fragment may migrate significantly cranially, caudally, or laterally from the disc of origin; the fragment is NOT visible at the level of the disc space on imaging (it has migrated) | A discrete free fragment of disc material within the epidural space, separate from the disc; T2 signal may be variable (low signal sequestra are desiccated and degenerate; high signal sequestra are hydrated and recent); no connection to the parent disc; may be found at a different level from the disc space | The most severe morphological type; the free fragment causes significant neural compression; surgical removal is typically required because spontaneous resorption of a completely sequestered fragment is less predictable; the fragment may compress the nerve root at an atypical level (above or below the disc of origin), explaining why symptoms may not match the expected dermatomal pattern for that disc level — `hidden` or migrated sequestra are a cause of failed conservative management and missed diagnoses on MRI |
Location of Herniation within the Canal
| Location | Description | Root Compressed | Clinical Effect |
|---|---|---|---|
| Posterolateral (paracentral) | The most common location (~90%); the herniation protrudes posterolaterally, into the lateral recess of the spinal canal; at this location it compresses the TRAVERSING (descending) nerve root — the root that is heading towards the foramen at the NEXT level below | At L4/5: compresses the L5 root (the traversing root); at L5/S1: compresses the S1 root; the `exiting` root at the level of herniation is spared in posterolateral herniations | Unilateral radiculopathy in the dermatomal distribution of the compressed root; the most common clinical presentation of disc herniation |
| Foraminal | The herniation occurs within the intervertebral foramen; compresses the EXITING (same-level) nerve root — the root that exits the foramen at the SAME level as the disc herniation | At L4/5: compresses the L4 root (the exiting root); at L5/S1: compresses the L5 root; foraminal herniations cause single-root compression at the SAME level rather than one level below | Severe radiculopathy; the compressed root is exiting the canal and has less space in the foramen than in the central canal; foraminal herniations are easily missed on MRI if the review is limited to axial sequences — dedicated foraminal sequences or careful sagittal review is needed |
| Far lateral (extraforaminal) | The herniation is entirely OUTSIDE the foramen — lateral to the pedicle; uncommon (~10%); often missed on standard MRI unless the field of view extends laterally enough | Compresses the EXITING root at that level; at L4/5: compresses L4; at L5/S1: compresses L5; same-level root compression — the symptoms match the SAME level disc, not one level below | Often requires a paramedian surgical approach (Wiltse approach) rather than the standard midline approach; can be missed unless specifically sought on imaging |
| Central | The herniation occurs centrally, compressing the central spinal canal; the thicker central PLL initially provides some protection; a large central herniation compresses the cauda equina (all the descending nerve roots) rather than a single root | The entire cauda equina is at risk; bilateral symptoms or saddle anaesthesia indicates central canal compromise; CAUDA EQUINA SYNDROME is the result of a large central herniation | SURGICAL EMERGENCY if cauda equina syndrome develops (bowel/bladder dysfunction, saddle anaesthesia, bilateral leg weakness); urgent MRI and surgical decompression |
Clinical Syndromes by Level
| Disc Level | Root Compressed (posterolateral) | Dermatomal Pain & Paraesthesia | Motor Weakness | Reflex Change | Clinical Test |
|---|---|---|---|---|---|
| L3/4 | L4 root | Anterolateral thigh, medial lower leg, medial malleolus (L4 dermatome); pain radiates from the back to the anterior thigh and medial shin | Quadriceps weakness (knee extension — L2, L3, L4); tibialis anterior weakness — foot dorsiflexion and inversion; difficulty walking on heels | Diminished or absent KNEE JERK (patellar reflex — L3, L4); the knee jerk is the most reliable reflex for L4 root compression | Femoral nerve stretch test (FNST) — patient prone, knee passively flexed — reproduces anterior thigh pain in L3/4 disc herniation; used for upper lumbar herniations (L1–L4) |
| L4/5 | L5 root | Posterior thigh, lateral lower leg, dorsum of foot and great toe (L5 dermatome — `lateral calf to big toe`) | Extensor hallucis longus (EHL) weakness — weakness of great toe dorsiflexion (most sensitive L5 motor sign); tibialis anterior weakness (foot drop — can walk on heels); gluteus medius weakness (Trendelenburg gait) | NO reliable reflex for L5 root alone; the posterior tibial reflex may be diminished but is unreliable clinically; the ABSENCE of a reflex change does NOT exclude L5 root compression | Straight leg raise (SLR) test positive at 30–60°; the most commonly involved root at the most common disc level; L4/5 disc herniations account for approximately 45–50% of all lumbar disc herniations |
| L5/S1 | S1 root | Posterior thigh, posterior calf, lateral border of foot, little toe (S1 dermatome — `lateral foot and little toe`); the classic `sciatica` dermatomal distribution | Gastrocnemius and soleus weakness — plantarflexion weakness (inability to stand on tiptoe on the affected side); peroneus longus weakness; gluteus maximus weakness; difficulty walking on tiptoes | Diminished or absent ANKLE JERK (Achilles reflex — S1, S2); the ankle jerk is the most reliable reflex for S1 root compression; asymmetric absence of the ankle jerk is highly specific for S1 root pathology | SLR positive; L5/S1 accounts for approximately 45–50% of all lumbar disc herniations; together L4/5 and L5/S1 account for 90–95% of all cases |
Clinical Assessment — Key Tests
- Straight Leg Raise (SLR — Lasègue test): the most important clinical test for lumbar disc herniation with root compression; performed with the patient supine — the hip is passively flexed with the knee straight; the test is POSITIVE when radicular pain (not just back pain or hamstring tightness) is reproduced in the dermatomal distribution of the affected root at 30–70° of hip flexion; the pain should be reproduced and should radiate below the knee; pain ONLY in the back is not a positive test; sensitivity ~80% (good for ruling out); specificity ~40% (poor for ruling in); the crossed SLR (raising the CONTRALATERAL leg reproduces ipsilateral radicular pain) is HIGHLY SPECIFIC (~90%) for a large central or paracentral disc herniation compressing the affected root from across the canal
- Bowstring sign (Cram test): with the SLR at the angle that reproduces symptoms, the knee is flexed slightly to relieve tension from the hamstrings — this should relieve the pain if it is due to hamstring tightness; then pressure is applied to the popliteal fossa (the sciatic nerve) — this reproduces the radicular pain if the SLR is a true positive; helps distinguish sciatic nerve tension from hamstring tightness
- Femoral nerve stretch test (FNST — reverse SLR): the patient is prone; the knee is passively flexed; this stretches the femoral nerve and the L2/3/4 roots; a positive test reproduces anterior thigh pain; used for UPPER lumbar disc herniations (L3/4 compressing L4; L2/3 compressing L3)
- Neurological examination must include: dermatomal sensory testing (light touch and pinprick); motor testing (key muscles — EHL for L5, plantarflexion for S1, quadriceps for L4); reflex testing (knee jerk L3/4; ankle jerk L5/S1 — note NO reliable reflex for isolated L5); gait assessment (foot drop = L5; inability to tiptoe = S1; Trendelenburg = L5 gluteus medius); assessment of sphincter function (anal tone, perianal sensation, bladder function) — any bowel or bladder dysfunction requires urgent assessment for cauda equina syndrome
- Waddell signs: five non-organic physical signs that may indicate a psychological or non-anatomical component to the patient`s symptoms; tenderness to superficial skin rolling; tenderness to axial loading (pressing on the crown of the skull reproduces back pain); simulated rotation; distracted SLR (positive when examined formally but negative when the patient is distracted — e.g. during hip examination with the knee extended); regional weakness or sensory disturbance in a non-dermatomal pattern; three or more positive Waddell signs suggest a significant non-organic component; should NOT be used to dismiss a patient`s symptoms without full investigation
Cauda Equina Syndrome — Surgical Emergency
- Cauda equina syndrome (CES): compression of the cauda equina (the descending lumbar and sacral nerve roots below the level of the conus medullaris — typically L1/2) causing a combination of lower limb neurological deficit and bowel/bladder dysfunction; most commonly caused by a large central lumbar disc herniation (especially L4/5); a SURGICAL EMERGENCY — delay in diagnosis and surgical decompression worsens long-term bladder and bowel outcomes
- Gleave-Macfarlane classification of CES: CES-I (incomplete) — urinary difficulties (hesitancy, altered urinary sensation, reduced stream) without complete retention; CES-R (retention) — complete urinary retention requiring catheterisation; CES-I has better prognosis than CES-R; outcome of surgical decompression is strongly correlated with the pre-operative neurological status — CES-I decompressed urgently has good prognosis; CES-R has poorer prognosis for complete recovery of bladder function even after decompression
- Cardinal features of CES: (1) Bilateral leg pain and weakness; (2) Saddle anaesthesia — loss of sensation in the perineum, scrotum/labia, inner thighs, and anal region — in the distribution of S3, S4, S5; (3) Bladder dysfunction — urinary retention (most common) OR incontinence (overflow); (4) Bowel dysfunction — faecal incontinence or constipation; (5) Sexual dysfunction — erectile dysfunction; ANY combination of these features with low back pain requires urgent MRI to exclude large central disc herniation
- Management of CES: URGENT MRI (the investigation of choice — demonstrates the level and severity of compression); if confirmed: URGENT surgical decompression (microdiscectomy or laminotomy and discectomy); timing of surgery is the subject of ongoing debate but current evidence supports surgery within 24–48 hours of onset of complete retention (CES-R); earlier decompression (within 24 hours) is associated with better neurological recovery in most case series; post-operative bladder function requires urodynamic monitoring; patients may require intermittent self-catheterisation for a variable period after surgery
Investigations
- MRI: the gold standard investigation for lumbar disc herniation; T2-weighted sequences demonstrate disc signal (low signal = degenerate; high signal = hydrated), herniation morphology and location, neural compression, and associated spinal stenosis; T1 sequences demonstrate bony anatomy and post-contrast enhancement (useful for distinguishing recurrent disc from epidural fibrosis after previous surgery); gadolinium contrast not routinely required for first-presentation disc herniation — use for post-operative patients or suspected infection/malignancy; MRI should NOT be performed for acute back pain without radicular features in the first 6 weeks (unless red flags are present) — it will detect incidental abnormalities that lead to unnecessary anxiety and treatment
- Plain radiographs: not diagnostic for disc herniation but useful for: demonstrating disc space narrowing (indirect evidence of disc degeneration); ruling out bony pathology (fracture, tumour, spondylolisthesis, infection); assessing alignment (scoliosis, kyphosis); dynamic (flexion-extension) views to assess instability; should be the first-line investigation in primary care before MRI
- CT myelography: the `gold standard` before MRI was available; now used when MRI is contraindicated (pacemaker, metallic implants) or when post-surgical scar tissue makes MRI interpretation difficult; CT without contrast provides excellent bony detail but poor soft tissue contrast for disc herniation
- Nerve conduction studies (NCS) and electromyography (EMG): used when the clinical presentation is atypical or when it is unclear whether symptoms are due to disc herniation, peripheral neuropathy, or plexopathy; EMG detects denervation (fibrillations, positive sharp waves) in the muscles supplied by the compressed root; NCS assesses peripheral nerve conduction; useful for establishing the chronicity of compression (acute vs chronic denervation patterns)
- Red flags requiring urgent investigation (NOT conservative management): progressive neurological deficit (worsening foot drop, increasing weakness); any features of cauda equina syndrome; history of malignancy; age >50 with new back pain; unexplained weight loss; fever; night sweats; pain worse at rest and at night; history of IV drug use; recent spinal procedure; these red flags indicate a potentially serious underlying cause (malignancy, infection, fracture) that requires urgent imaging and investigation before assuming the cause is degenerative disc disease
Conservative Management
- Natural history of lumbar disc herniation: approximately 90% of patients with lumbar radiculopathy from disc herniation will improve significantly or completely with conservative management within 6–12 weeks; the natural history of disc herniations is favourable because: (1) the herniated fragment undergoes spontaneous resorption over weeks to months — extrusions and sequestrations have the fastest resorption rates (exposure of nucleus material to the epidural space triggers an inflammatory response with macrophage-mediated phagocytosis of the disc material); (2) the inflammatory mediators that cause neural sensitisation (prostaglandins, cytokines, phospholipase A2) dissipate over time; this favourable natural history means that the primary goal of conservative management is to MANAGE SYMPTOMS while awaiting spontaneous resolution, NOT to prevent resolution
- Analgesia: a stepwise approach — paracetamol (insufficient evidence for efficacy in radicular pain but safe and widely used); NSAIDs (ibuprofen, naproxen, diclofenac) — moderate evidence for reduction of pain; short-course oral steroids (dexamethasone or prednisolone) — evidence for short-term improvement in acute severe radiculopathy; neuropathic agents (pregabalin, gabapentin, amitriptyline) — for neuropathic pain (burning, paraesthesia); strong opioids — reserved for severe acute pain; not recommended for long-term management of radiculopathy due to dependency risk and evidence of no long-term benefit over placebo
- Physiotherapy: active exercise and physiotherapy is superior to bed rest (which worsens outcomes) and passive treatments; evidence supports: gradual return to activity; McKenzie technique (extension exercises for posterior disc herniation); core stabilisation exercises; walking programme; physiotherapy does not alter the disc herniation itself but improves functional recovery and reduces disability; activity modification (avoiding heavy lifting, prolonged sitting, bending, twisting) reduces symptoms during the acute phase
- Epidural steroid injections (ESI): transforaminal or interlaminar ESI delivers corticosteroid to the epidural space adjacent to the compressed nerve root; evidence: short-term benefit (4–6 weeks) for radicular pain and function; no evidence of long-term benefit over placebo at 12 months; does NOT reduce the rate of eventual surgery; indicated for: failure of oral analgesia and physiotherapy; while awaiting surgical assessment; when surgery is contraindicated; the SPORT trial showed no significant difference in surgical vs non-surgical outcomes for disc herniation at 4 years — suggesting many surgically treated patients would have improved with continued conservative management
Surgical Management — Microdiscectomy
- Indications for surgery: (1) ABSOLUTE indication — cauda equina syndrome (urgent surgery); progressive neurological deficit (foot drop, rapidly worsening weakness); (2) RELATIVE indications — failure of conservative management at 6 weeks for incapacitating radiculopathy; persistent significant neurological deficit at 6 weeks despite conservative management; patient preference after full discussion of the evidence (surgery provides faster pain relief but equivalent outcomes at 1–2 years compared to continued conservative management — the SPORT trial); clinically and radiologically concordant findings (symptoms and imaging must match — the WRONG root is decompressed if they do not)
- Microdiscectomy: the gold standard surgical procedure for lumbar disc herniation; performed under general anaesthesia in the prone position; a 2–3 cm paramedian skin incision; subperiosteal dissection of the paraspinal muscles; partial laminotomy (removal of the inferior edge of the lamina above and the superior edge of the lamina below the disc space — a `hemilaminotomy`); ligamentum flavum excision; identification and gentle retraction of the traversing nerve root medially; removal of the herniated disc material (both the accessible herniation AND residual disc material from within the disc space using pituitary rongeurs and curettes); haemostasis; closure in layers; the use of an operating microscope or loupes provides magnification and illumination to minimise neural trauma
- Outcomes: 80–90% of patients report significant improvement in leg pain after microdiscectomy; results for back pain are less predictable (surgery addresses the radiculopathy, not the degenerative disc disease that caused it); the SPORT RCT (Weinstein et al., NEJM 2006) showed that both surgical and non-surgical patients with disc herniation improved significantly at 1 and 2 years, but surgically treated patients had faster and greater short-term relief of leg pain; at 4 years, outcomes were similar between groups
- Macnab criteria for outcome assessment after disc surgery: EXCELLENT — complete relief of all pre-operative symptoms; GOOD — occasional non-limiting pain but no restriction of activities; FAIR — some improvement but residual pain affecting activities (restriction of ADL and/or recreational activities); POOR — no improvement OR worse than pre-operatively; Macnab criteria are the most widely quoted outcome classification for lumbar disc surgery in the literature; excellent/good outcomes together are reported in approximately 85–90% of patients at 1 year after microdiscectomy
- Complications of microdiscectomy: dural tear (1–5% — intraoperative repair with direct suture or dural patch; post-operative CSF leak if unrecognised); nerve root injury (<1% — most commonly from aggressive retraction); disc space infection / discitis (1–2%); recurrent disc herniation at the same level (5–15% — the most common cause of failed back surgery after microdiscectomy; presents with recurrence of the pre-operative radiculopathy; managed conservatively first; re-operation rates are lower than primary surgery outcomes); wrong-level surgery (a `never event` — prevented by intraoperative fluoroscopic level confirmation); haematoma
Exam Pearls
- Disc herniation morphology: protrusion (base wider than height — annulus partially intact); extrusion (height wider than base — nucleus through full annulus, still connected); sequestration (free fragment — no connection to parent disc); bulge = NOT a herniation (generalised, >25% circumference)
- Location and root: posterolateral herniation = TRAVERSING root (one level below); foraminal/far lateral = EXITING root (same level); L4/5 posterolateral = L5 root; L5/S1 posterolateral = S1 root; central = cauda equina
- Level-specific syndromes: L4 root (L3/4 disc) — weak quads, knee jerk reduced, anterior thigh/medial shin pain; L5 root (L4/5 disc) — weak EHL (big toe extension), NO reflex change (classic exam point), lateral calf/dorsum foot pain, foot drop in severe cases; S1 root (L5/S1 disc) — weak plantarflexion, ankle jerk reduced, posterior calf/lateral foot/little toe pain
- SLR: sensitivity 80% (good for ruling out); specificity 40% (poor for ruling in); positive at 30–70° — reproduces radicular pain BELOW THE KNEE; back pain alone is NOT a positive SLR; crossed SLR specificity ~90% (highly specific for large central or paracentral disc)
- Cauda equina syndrome: SURGICAL EMERGENCY; large central disc herniation; saddle anaesthesia; bilateral leg weakness; urinary retention (CES-R) or dysfunction (CES-I); Gleave-Macfarlane: CES-I (incomplete) = better prognosis; CES-R (retention) = worse; urgent MRI → surgery within 24–48 hours
- Natural history: 90% resolve conservatively within 6–12 weeks; extrusions and sequestrations resorb FASTER than protrusions (greater inflammatory response); conservative management = analgesia + physiotherapy + activity modification; NSAIDs; epidural steroid injections for short-term relief; no evidence of long-term benefit over placebo at 12 months
- Microdiscectomy indications: cauda equina syndrome (absolute, urgent); progressive neurological deficit (absolute); failure of 6 weeks conservative management for incapacitating radiculopathy (relative); SPORT trial — surgery provides faster relief but similar outcomes to conservative management at 4 years; Macnab outcome criteria: excellent (complete relief); good (occasional non-limiting pain); fair (some improvement, residual restriction); poor (no improvement or worse)
- Recurrent disc herniation: 5–15% rate after microdiscectomy; same level, same or contralateral side; most common cause of failed back surgery syndrome; manage conservatively first; re-operation has slightly lower success rates than primary surgery